|
We present a signature of 33 distinct pathophysiological proteoforms, including the commonly targeted Aβ 40, Aβ 42, Aβ 4-42, Aβ 11-42, and provide insight into their synthesis and quantities. Interactome analysis was performed by co-immunoprecipitation. The biophysical analysis was conducted using RT-QuIC assay, confocal microscopy, and atomic force microscopy. 
We employed two-dimensional polyacrylamide gel electrophoresis and MALDI-ToF mass spectrometry to validate and identify the Aβ proteoforms extracted from targeted brain tissues. The current study was designed to identify these variations in Alzheimer’s disease patients exhibiting classical (sAD) and rapid progression, with the primary aim of establishing if these variants may constitute strains that underlie the phenotypic variability of Alzheimer’s disease. 
The Aβ peptide can exist as multiple proteoforms that vary in their post-translational processing, amyloidogenesis, and toxicity. Fibrilization of the amyloid-β (Aβ) peptide is the most frequently studied candidate in this context.  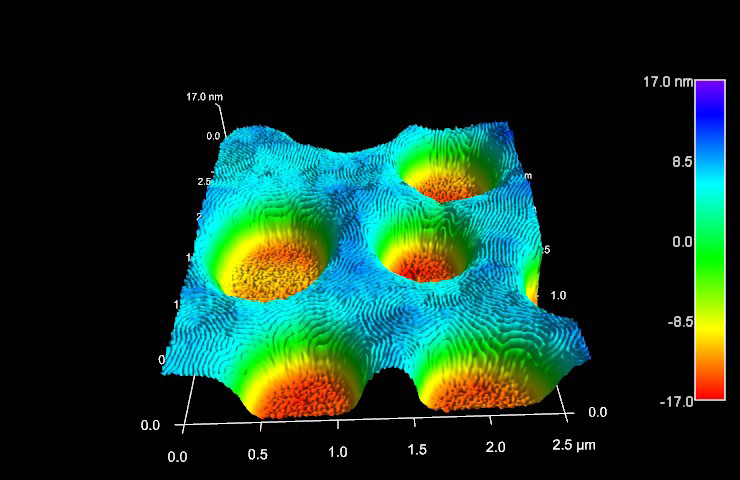
The molecular determinants of atypical clinical variants of Alzheimer’s disease, including the recently discovered rapidly progressive Alzheimer’s disease (rpAD), are unknown to date.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
